All published articles of this journal are available on ScienceDirect.
Protective Role of Aframomum melegueta against Monosodium Glutamate-induced Prostate Damage in Rats
Abstract
Introduction
Prostate cancer is among the most prevalent cancers in men, with increasing incidence, and the limited therapeutic options have stimulated interest in plant-based bioactive compounds as alternatives. Aframomum melegueta is widely utilized in ethnomedicine for various health issues and has drawn interest due to its promising therapeutic properties. This study investigated the protective and therapeutic role of ethanol seed extract of Aframomum melegueta (ESEAM) against MSG-induced prostate damage in rats, integrating in vivo experimental evidence with in silico analysis to establish its relevance in prostate-related drug discovery.
Methods
Male rats were divided into groups and induced with MSG with or without treatment using ESEAM or finasteride. Biochemical assays and histopathological evaluation were conducted to assess protective and restorative effects. Molecular docking was performed to predict interactions between phytochemicals and prostate damage-associated protein targets. Data were presented as mean ± SEM and analyzed using one-way ANOVA followed by Tukey’s post-hoc test (p < 0.05).
Results
MSG exposure induced significant alterations in hematological and biochemical indices, with severe histopathological alterations. Compared with the control, ESEAM significantly (p < 0.05) improved hematological indices and lowered liver and prostate markers, with histology confirming notable attenuation of prostate damage. Molecular docking revealed favorable binding interactions between ESEAM phytochemicals (552098, 296573, 5364759, and 296573) to prostate-related molecular targets.
Discussion
The observed biochemical, histological, and computational findings suggest that ESEAM exerts protective and restorative effects against MSG-induced prostate injury, possibly through bioactive compound–target interactions that modulate prostate-related pathways.
Conclusion
Collectively, these findings indicate that ESEAM displays protective and therapeutic effects and represents a promising drug candidate for further mechanistic and drug development.
1. INTRODUCTION
Prostate cancer and other related prostate damage are the significant forms of malignancy in men, especially in developed countries, where most of the cases are diagnosed in men above 50 years of age [1]. There are no known causes of prostate cancer, but reports suggest that dairy factors are major risk determinants [2-4]. Hormones, mainly androgens and estrogens, have been highly involved in prostate carcinogenesis as well, but evidence from human models is also lacking. Nonetheless, the scientific explanation of prostate cancer biology suggests that androgen and possibly estrogen may be responsible. Infections (prostatitis, inflammation of the prostate) are also considered prostate cancer risk factors [5]. In addition, studies from experimental animals have shown that chemical agents, most especially those metabolically activated in the prostate, can trigger prostate carcinogenesis [6].
The sodium salt of glutamic acid, monosodium glutamate (MSG), dissociates in water to yield free sodium ions and glutamic acid [7], which stimulates synaptic receptors (glutamate receptors) on neuronal membranes and plays a central role in excitotoxicity. Its overstimulation by MSG can trigger signaling pathways linked to cell survival and proliferation [8], potentially mimicking mechanisms seen in neurological disorders, where glutamate excitotoxicity causes cellular stress. The toxic effects of MSG on the testis cause significant oligozoospermia and an increase in sperm morphology in a dose-dependent fashion in male Swiss albino rats [9, 10]. It has also been implicated in causing degeneration, testicular hemorrhage, alteration of sperm cell morphology, and population in male infertility [8, 11]. Furthermore, overuse of MSG as a flavor enhancer has been reported to be hepatotoxic [12]. In contrast, in the animal model, chronic MSG intake has been reported to induce kidney damage via oxidative stress [13, 14], and ROS formation, both of which contribute significantly to its nephrotoxic effect, thereby causing cellular and functional damage [15]. These suggest MSG can be a useful experimental model for studying chemically induced prostate damage.
Hormone therapy, radiation, surgical treatment, and chemotherapy are the currently available treatment options for prostate cancer and or damage. These treatments have shown remarkable improvement in patients, but have come up with adverse and toxic side effects, creating a need for alternative therapy with improved efficacy and safety profiles [15, 16]. Among available therapies, finasteride is widely used as a reference drug in both human and animal models. It is a selective 5α-reductase inhibitor that prevents the conversion of testosterone to dihydrotestosterone (DHT), thereby suppressing androgen-driven prostate growth and carcinogenesis. It has been clinically approved for benign hyperplasia and investigated in prostate cancer chemoprevention trials [17]. The use of natural and dietary supplements, such as vitamin E, selenium, lycopene, and green tea, is widely accepted in prostate cancer treatment [18-21]. According to the World Health Organization, about 65% of the global population feels safe with treating disease(s) with traditional medicine [22, 23] with India, China, and Africa, along with the USA, having an increased usage of complementary and alternative drugs within the last 2 decades [24, 25]. Approximately 80% of anticancer agents are derived from plants and other natural resources; however, they have not been explored in the management of prostate cancer or prostate-related damage [26-28].
Aframomum melegueta is a Zingiberaceae family plant spice widely cultivated in Africa for its valuable seeds. It is an herbaceous perennial plant native to swampy habitats along the West African coast. It is commonly known as “grains of paradise,” “melegueta pepper,” “alligator pepper,” “guinea grains,” or “guinea pepper” [29, 30]. Apart from their culinary uses, the seeds of A. melegueta are claimed to have antioxidant [31], anti-cancer [32-34], anti-diabetic [35, 36], anti-inflammatory [37], and hepatoprotective activity [38]. Okeke et al. [39] reported that the seed of A. melegueta helps in improving penile rigidity and erectile function. Despite the numerous therapeutic applications and scientific reports on this plant, there is no report on the effectiveness of this plant on prostate cancer or prostate-related damage with respect to prostate markers in silico. Given the limitations, we used this plant to evaluate its therapeutic potential for treating prostate damage using experimental and computational approaches.
2. MATERIALS AND METHODS
2.1. Chemicals
Monosodium glutamate and finasteride were purchased from Sigma Chemical Company (St. Louis, MO, USA). Prostate-specific antigen (PSA) and luteinizing hormone (LH) ELISA assay kit (Calbiotech Inc., CA, USA), interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) ELISA assay kit (Bioassay Tech. Lab. Shanghai, China). All other chemicals and solvents used were of analytical grade and the highest purity.
2.2. Collection and Authentication of the Plant Part
A. melegueta seeds were collected from Oja Oba Market in Ilorin, Kwara State. It was identified and authenticated at the Herbarium Unit of the Department of Plant Biology, University of Ilorin, where a voucher number, UILH/001/2019/1166, was assigned and deposited in the University Herbarium. Prior to extraction, the shells of A. melegueta seeds were removed and ground into a fine powder (KitchenAid KSB4027PA Countertop Blender, USA). About 3 kg of ground seed was soaked in 3 L of 98% ethanol for 24 hours, then filtered. The ethanol filtrate was concentrated under reduced pressure using a rotary evaporator. The resulting residue, called ethanol seed extract of Aframomum melegueta (ESEAM), was transferred to a hot-air oven, where it was dried to constant weight at 40ºC, stored in air-tight sample bottles, and kept until used.
2.3. Phytochemical and GC-MS Analysis of ESEAM
The phytochemical profile of ESEAM has been previously reported in our earlier study (Table 1) [40]. In brief, qualitative phytochemical screening revealed the presence of alkaloids, flavonoids, phenols, saponins, and tannins. At the same time, gas chromatography-mass spectrometry (GC-MS) analysis further identifies some bioactive compounds, notably, caryophyllene, humulene, 5α-Androstan-16-one, and other lipid compounds. For the current study, the same extraction procedure was adopted to ensure reproducibility. The previously identified phytochemical constituents were also considered as putative ligands for in silico docking against enzymes implicated in prostate damage.
| S.No. | RT (min) | Name of Compound | MW (g/mol) | Peak Area (%) | Peak Height (%) |
|---|---|---|---|---|---|
| 1 | 5.144 | 2-Nonanol | 144.25 | 0.35 | 0.42 |
| 2 | 5.374 | 1-Octene, 3-(methoxymethoxy) | 172.26 | 2.96 | 1.16 |
| 3 | 6.120 | 2-Heptanol, acetate | 158.24 | 0.57 | 1.04 |
| 4 | 6.981 | 1,6-Octadien-3-ol, 3,7-dimethyl | 194.31 | 0.72 | 1.84 |
| 5 | 8.183 | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | 160.12 | 6.44 | 1.73 |
| 6 | 8.706 | 2-Butanone, 4-hydroxy-3-methyl | 178.23 | 2.53 | 0.91 |
| 7 | 9.021 | Thiophene, 2-propyl | 126.22 | 1.14 | 0.69 |
| 8 | 9.583 | Cycloheptane, 1,4-dimethoxy-, trans | 158.24 | 0.11 | 2.20 |
| 9 | 9.812 | Urea, 1-(p-nitrobenzoyl)-3-phenyl | 285.25 | 0.42 | 0.49 |
| 10 | 10.593 | Diethylene glycol monododecyl ether | 274.44 | 0.44 | 0.35 |
| 11 | 10.894 | 8-Methyl-6-nonenoic acid | 170.25 | 0.61 | 0.63 |
| 12 | 11.462 | Caryophyllene | 204.35 | 5.00 | 7.82 |
| 13 | 11.830 | Humulene | 204.35 | 7.25 | 9.87 |
| 14 | 12.093 | 5α-Androstan-16-one | 290.4 | 0.09 | 0.39 |
| 15 | 12.376 | Toluene-4-sulfonic acid, 2,7-dioxatricyclo[4.3.1.0(3,8)]dec-10-yl | 310.4 | 0.25 | 1.00 |
| 16 | 12.681 | Caprylic anhydride | 270.41 | 2.44 | 1.36 |
| 17 | 12,965 | cis-Z-α-Bisabolene epoxide | 220.35 | 0.23 | 1.26 |
| 18 | 13.187 | 12-Oxabicyclo[9.1.0]dodeca-3,7-dien | 220.35 | 0.44 | 1.95 |
| 19 | 14.346 | Octadecanoic acid | 284.5 | 1.75 | 6.98 |
| 20 | 15.071 | Pentadecanoic acid | 242.4 | 7.69 | 1.80 |
| 21 | 15.962 | n-Hexadecanoic acid | 256.42 | 11.32 | 6.98 |
| 22 | 16.390 | Cyclopentadecanone, 2-hydroxy | 240.38 | 2.59 | 1.78 |
| 23 | 17.039 | 9,12-Octadecadienoic acid (Z,Z) | 296.4 | 13.18 | 7.07 |
| 24 | 17.205 | Octadecanoic acid | 284.5 | 0.55 | 2.50 |
| 25 | 17.248 | Ethyl 14-methyl-hexadecanoate | 298.5 | 0.23 | 1.19 |
| 26 | 17.416 | Naphthalene, 2-decyldecahydro | 278.5 | 0.38 | 1.16 |
| 27 | 17.772 | Naphthalene, decahydro-1-pentadecyl- | 348.6 | 2.62 | 2.46 |
| 28 | 17.904 | [1,3]Benzodioxolo[5,6-c]phenanthridine, 12,13-dihydro-2,3-dim | 712.7 | 3.06 | 2.53 |
| 29 | 18.746 | Hexadecanal, 2-methyl | 254.5 | 2.98 | 2.05 |
| 30 | 19.133 | Diisooctyl phthalate | 390.6 | 1.54 | 1.61 |
| 31 | 19.452 | Ethyl 9-hexadecenoate | 282.5 | 1.39 | 1.20 |
| 32 | 19.957 | (R)-(-)-14-Methyl-8-hexadecyn-1-ol | 252.4 | 1.97 | 4.88 |
| 33 | 20.072 | Octanoic acid, 2,3-dihydroxypropyl ester | 218.29 | 0.61 | 2.10 |
| 34 | 21.056 | E-11-Tetradecen-1-ol difluoroacetate | 290.39 | 0.66 | 0.81 |
| 35 | 21.558 | d-Norandrostane(5α,14α) | 246.4 | 1.53 | 2.49 |
Abbreviations: RT = Retention time: MW = Molecular weight.
2.4. Animal Studies
Adult male Swiss albino rats (6 weeks old) weighing 200.09 ± 2.20 g were purchased from the Animal Holding Unit of the Department of Biochemistry, University of Ilorin, Ilorin, Nigeria, and acclimatized for 5 days. Castration was performed according to the method of Dulisch [41], and the rats were left to recover for another week, with constant monitoring for signs of pain, distress, or illness and humane endpoints daily, in accordance with the guidelines of the Animal Care and Use Committee of Kwara State University. Animals were then completely randomized to five (5) groups of ten animals each. All rats were housed in a pathogen-free environment and provided ad libitum access to food and water. Our methods were carried out in accordance with international guidelines and regulations on ethical principles for research using laboratory animals, adhering to the principles of the 3Rs (US NRC’s guide). The study was approved and conducted in accordance with the ethical procedures and policies of the Animal Care and Use Committee of Kwara State University, Malete, Nigeria, and an approval number (KWASU/CR&D/REA/2024/050) was assigned.
2.5. Sensitization and Treatment
Seventy (70) male rats were castrated and allowed one week of recovery before experimentation. Following castration, fifty (50) rats (200.09 ± 2.20 g) were divided into five groups (n=10) and used for the study. Animals were excluded from the study if they exhibited post-surgical complications, poor recovery following castration, signs of infection, abnormal behavior, or failure to meet the predefined health and physiological criteria required for inclusion. Following the recovery period, only animals that demonstrated complete healing and stable physiological conditions were selected for the study. Consequently, out of the initial 70 mice, 50 met the inclusion criteria and were included in the final experimental analysis. Except for the control group, all rats received 60 mg/kg body weight MSG intraperitoneally in 2 mL distilled water for 28 days to induce and promote prostate damage. Control rats were administered distilled water alone. ESEAM or finasteride was administered to the sensitized rats at 50 mg/kg, 100 mg/kg, or 0.8 mg/kg body weight, respectively, for 28 days. During the experiment, mortality was recorded in the MSG-only group, resulting in a reduction in the number of animals available for analysis. On the 28th day, all animals were sacrificed under light anesthesia performed as described previously by Ku et al [42], and blood and tissue samples were collected for biochemical estimations and histopathological examination.
2.6. Biochemical Analysis
Whole blood samples were collected from rats by cardiac puncture under anesthesia into EDTA sample bottles for hematological studies. At the same time, serum was extracted from whole blood by centrifugation at 5000 rpm using a bench-top centrifuge (Frontier 500 series). Liver, kidney, and prostate tissues were collected and perfused immediately with 0.25 M ice-cold sucrose solution. They were homogenized in sucrose buffer (pH 7.4) to yield a 10% (w/v) homogenate, then centrifuged at 10,000 g for 20 mins. The supernatant (cytosol fraction), after discarding any floating lipid layer and appropriate dilution, was used to assay biochemical analysis. An autohematological analyzer (Shenzhen Mindray, BioMedical Electronics Co., Ltd., China) was used to study hematological indices. AST, ALT, GGT, and ALP were measured using Randox kits (Randox Laboratories Ltd., Antrim, UK). Urea determination was based on the cleavage of urea with urease (Berthelot's reaction) according to Fawcett and Scott [43]. Uric acid was determined using the phosphotungstate method as described by Itiaba [44], and creatinine was measured using Jaffe’s modified method [45]. Prostate-Specific Antigen (PSA) and Lutenizing Hormone (LH) were estimated using an ELISA assay kit (Calbiotech Inc., CA, USA), total acid phosphatase (TACP) and prostatic acid phosphatase (PACP) were assessed as described by Mahmud et al. [46], interleukin-6 (IL-6) and tumor necrosis factor-α (TNF- α) were estimated using an ELISA assay kit (Bioassay Tech. Lab. Shanghai, China).
2.7. Histology
The prostate was collected from euthanized animals and was fixed in 10% paraformaldehyde (PFA), which was routinely processed and paraffin-embedded. This was done according to the method of Ramachandran and Kakar [47].
2.8. Molecular Docking
2.8.1. Protein Retrieval and Preparation
The target proteins with PDB IDs: 1ANI, 1IVR, 3IJH, and 4Z9O for alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, and gamma-glutamyl transpeptidase 1, respectively, were downloaded from the RSCB Protein Data Bank [48, 49]. All the respective PDB IDs were uploaded to the Molegro Virtual Docker (MVD) 2013 V6.0. Water molecules, cofactors, ligands, and other non-essential molecules in the crystal structures were removed, and protein structural defects were identified for each of the four enzymes. Structural defects in amino acid residues were examined, corrected, and optimized using neighboring residues. The surface was generated for each protein, and a ligand-binding cavity was identified separately for each enzyme after adding hydrogen atoms and assigning charges. Each enzyme was set at a different coordinate along the X, Y, and Z axes. The X, Y, and Z axes were set at 62.78, 31.26, and 26.58 for 1ANI, 46.94, 12.95, and 39.21 for 1IVR, -17.26, 59.32, and 14.00 for 3IJH, -20.80, -26.16, and -10.90 for 4Z9O. Grid resolution was set to 0.30 Armstrong for all PDB IDs, and the maximum energy was minimized.
2.8.2. Ligand Preparation
A total of 30 ligands were downloaded from PubChem in the form of 3D SDF conformers, based on the results of our GC-MS analysis of A. melegueta (Table 1) [40]. Finasteride was used as the reference drug for the study. All ligands were uploaded to MVD and prepared to correct any errors present. The preparation options were set to “if missing” for assign bonds, hybridization, create explicit hydrogens, assign tripos atom types, create explicit hydrogens, and assign charges. This was achieved using the Molegro Virtual Docker (MVD) software V2013. Ligands were then docked to each receptor using the Dock Wizard option to obtain docking scores.
2.9. Statistical Analysis
Values were presented as mean ± SEM. The normality and homogeneity of variance were assessed using one-way analysis of variance, followed by Tukey’s post-hoc test for multiple comparisons with variables that showed significant differences (p < 0.05) between the groups. All statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) version 16 software (Chicago, IL, USA).
3. RESULTS
3.1. Effect of ESEAM on Hematological Parameters
The distribution of some hematological parameters (Table 2) for MSG-induced prostate damage was evaluated. It revealed a significant decrease (p < 0.05) in the MSG-treated group compared with the control, and upon treatment with the extract, a substantial increase was observed.
| Enzymes/Organs | Control | MSG (60 mg/kg) | ESEAM (50 mg/kg) | ESEAM (100 mg/kg) | Finasteride (0.8 mg/kg) |
|---|---|---|---|---|---|
| Red blood cell (x1012/l) | 4.83 ± 0.42b | 3.58 ± 0.89a | 4.98 ± 0.46c | 4.72 ± 0.59b | 4.79 ± 0.56b |
| Hemoglobulin (g/dl) | 7.84 ± 0.59b | 6.82 ± 1.72a | 9.62 ± 0.88d | 9.22 ± 1.16c | 9.26 ± 1.01c |
| Packed cell volume (%) | 27.20 ± 2.27c | 19.80 ± 5.01a | 27.20 ± 2.31c | 27.20 ± 3.71c | 26.40 ± 3.23b |
| Mean cell volume | 55.80 ± 0.97c | 44.40 ± 11.10a | 56.40 ± 2.79d | 56.80 ± 2.78e | 54.40 ± 1.63b |
| Mean cell hemoglobin (%) | 19.04 ± 0.22b | 15.24 ± 3.81a | 19.28 ± 0.29d | 19.22 ± 0.19d | 19.32 ± 0.26c |
| Mean cell hemoglobin concentration (%) | 33.88 ± 0.48b | 27.20 ± 6.82a | 34.54 ± 1.07c | 35.80 ± 1.41d | 35.44 ± 1.40d |
| White blood cell count (x109/L) | 6.52 ± 2.12b | 8.46 ± 0.98c | 6.03 ± 1.09a | 6.03 ± 1.09a | 6.47 ± 0.97b |
| Neutrophils (%) | 32.80 ± 3.48c | 23.00 ± 5.81a | 38.40 ± 3.39e | 35.80 ± 3.99d | 29.40 ± 1.75b |
| Lymphocytes (%) | 63.40 ± 3.88e | 56.40 ± 14.12a | 60.60 ± 3.71c | 62.80 ± 4.27d | 58.80 ± 14.76b |
| Monocytes (%) | 0.00 ± 0.00a | 1.40 ± 0.51d | 1.00 ± 0.45c | 0.80 ± 0.37b | 0.80 ± 0.37b |
| Eosinophils (%) | 0.00 ± 0.00a | 0.40 ± 0.24b | 0.00 ± 0.00a | 0.00 ± 0.00a | 0.00 ± 0.00a |
| Basophils (%) | 0.00 ± 0.00a | 0.20 ± 0.20b | 0.00 ± 0.00a | 0.00 ± 0.00a | 0.00 ± 0.00a |
| Platelet (x109/l) | 202.20 ± 11.88d | 161.60 ± 40.51a | 192.80 ± 19.04b | 212.80 ± 9.39e | 201.00 ± 13.96c |
Note: Values are expressed as mean ± SEM, n = 6 rats. Analysis of variance (p < 0.05).
3.2. Effect of ESEAM on Serum and Liver Markers and Kidney Indices
Administration of MSG induced hepatotoxicity by increasing significantly (p < 0.05) serum and liver activities of AST, ALT, ALP, and GGT in the MSG-only group compared to the control group. Treatment with the ESEAM at doses of 50 and 100 mg/kg body weight, respectively, induced a significant reduction (p < 0.05) (Table 3). Similarly, there was a substantial increase (p < 0.05) in urea, uric acid, and creatinine in animals treated with MSG. Still, upon treatment with ESEAM, there was a significant decrease (p < 0.05) in the level of kidney indices (Table 4).
| Enzymes (U/L)/Organs | Control | MSG (60 mg/kg) | ESEAM (50 mg/kg) | ESEAM (100 mg/kg) | Finasteride (0.8 mg/kg) |
|---|---|---|---|---|---|
| ALP | - | - | - | - | - |
| Serum | 81.87 ± 1.29a | 103.00 ± 0.72e | 96.63 ± 0.46d | 88.63 ± 0.59c | 81.70 ± 0.63b |
| Liver | 81.57 ± 0.67a | 108.73 ± 3.10e | 93.80 ± 1.04d | 85.33 ± 0.58b | 87.63 ± 0.46c |
| AST | - | - | - | - | - |
| Serum | 36.03 ± 0.30a | 61.70 ± 0.70e | 56.67 ± 2.40d | 49.70 ± 0.50c | 45.60 ± 0.60b |
| Liver | 51.27 ± 0.72a | 68.13 ± 1.27e | 57.70 ± 1.36d | 47.40 ± 0.64c | 43.87 ± 0.35b |
| ALT | - | - | - | - | - |
| Serum | 18.70 ± 0.10a | 41.23 ± 0.50e | 35.40 ± 0.20d | 27.90 ± 0.27c | 25.90 ± 0.28b |
| Liver | 17.83 ± 0.35a | 41.80 ± 0.58e | 38.00 ± 0.26d | 30.67 ± 0.41c | 28.20 ± 0.47b |
| GGT | - | - | - | - | - |
| Serum | 20.76 ± 0.29a | 30.86 ± 0.20e | 23.10 ± 0.34d | 17.73 ± 0.27c | 15.93 ± 0.18b |
| Liver | 36.17 ± 0.41c | 62.37 ± 0.39e | 41.50 ± 0.64d | 23.70 ± 1.46a | 26.30 ± 0.22b |
Note: Values are expressed as mean ± SEM, n = 6 rats. Analysis of variance (p < 0.05).
| Enzymes/Organs | Control | MSG (60 mg/kg) | ESEAM (50 mg/kg) | ESEAM (100 mg/kg) | Finasteride (0.8 mg/kg) |
|---|---|---|---|---|---|
| Urea (mg/dl) | - | - | - | - | - |
| Serum | 2.70 ± 0.05a | 4.77 ± 0.08e | 3.53 ± 0.03d | 3.33 ± 0.12c | 3.10 ± 0.06b |
| Kidney | 2.50 ± 0.06c | 4.10 ± 0.12e | 3.67 ± 0.09d | 2.23 ± 0.03b | 2.13 ± 0.09a |
| Uric Acid (mg/dl) | - | - | - | - | - |
| Serum | 0.13 ± 0.02a | 0.36 ± 0.01c | 0.23 ± 0.01b | 0.18 ± 0.01a | 0.17 ± 0.01a |
| Kidney | 0.15 ± 0.02a | 0.38 ± 0.01d | 0.31 ± 0.01bc | 0.27 ± 0.01b | 0.23 ± 0.01b |
| Creatinine (mmol/l) | - | - | - | - | - |
| Serum | 55.03 ±0.38a | 80.58 ± 0.61e | 76.20 ± 1.35d | 60.89 ± 0.29b | 62.37 ± 0.58c |
| Kidney | 51.70 ±0.74a | 82.23 ± 0.82e | 69.53 ± 0.68d | 57.20 ± 0.51c | 52.53 ± 0.52b |
Note: Values are expressed as mean ± SEM, n = 6 rats. Analysis of variance (p < 0.05).
3.3. Effect of ESEAM on Serum and Prostate Markers
The relative serum and tissue activities of prostate markers (PSA, LH, TACP, and PACP) were elevated (p < 0.05) in animals treated with MSG when compared with the control (Table 5). Upon treatment with the extract, a significant reduction (p < 0.05) was observed.
| Enzymes/Organs | Control | MSG (60 mg/kg) | ESEAM (50 mg/kg) | ESEAM (100 mg/kg) | Finasteride (0.8 mg/kg) |
|---|---|---|---|---|---|
| PSA (ŋg/mL) | - | - | - | - | - |
| Serum | 0.35 ± 0.03a | 6.48 ± 0.21d | 0.88 ± 0.06c | 0.85 ± 0.05c | 0.57 ± 0.13b |
| Prostate | 0.34 ± 0.01a | 3.87 ± 0.42e | 0.59 ± 0.04c | 0.67 ± 0.04d | 0.49 ± 0.06b |
| LH (IU/L) | - | - | - | - | - |
| Serum | 0.11 ± 0.01a | 0.38 ± 0.02c | 0.18 ± 0.01ab | 0.13 ± 0.01a | 0.13 ± 0.02a |
| Prostate | 0.13 ± 0.01a | 0.47 ± 0.02c | 0.19 ± 0.00ab | 0.17 ± 0.01a | 0.19 ± 0.00ab |
| TACP (ŋg/mL) | - | - | - | - | - |
| Serum | 5.76 ± 0.04a | 17.72 ± 1.28e | 9.18 ± 0.60d | 7.20 ± 0.37c | 6.13 ± 0.62b |
| Prostate | 6.41 ± 0.71a | 12.66 ± 0.56d | 8.20 ± 0.37c | 6.72 ± 0.72b | 6.41 ± 0.71a |
| PACP (ŋg/mL) | - | - | - | - | - |
| Serum | 2.07 ± 0.09a | 8.76 ± 0.13e | 7.70 ± 0.78d | 4.62 ± 0.04c | 4.12 ± 0.28b |
| Prostate | 2.95 ± 0.04a | 12.05 ± 0.13e | 5.38 ± 0.21d | 3.27 ± 0.06c | 3.03 ± 0.12b |
Note: Values are expressed as mean ± SEM, n = 6 rats. Analysis of variance (p < 0.05).
Abbreviations: PSA = Prostate-specific antigen; LH = Luteinizing hormone; TACP = Total acid phosphatase; PACP = Prostate acid phosphatase.
3.4. Effect of ESEAM on Prostate Tissue Histopathology in MSG-induced Prostate Damage in Rat
The effect of ESEAM on prostate tissue histology in MSG-induced prostate-damaged rats is shown in Fig. (1a-e).
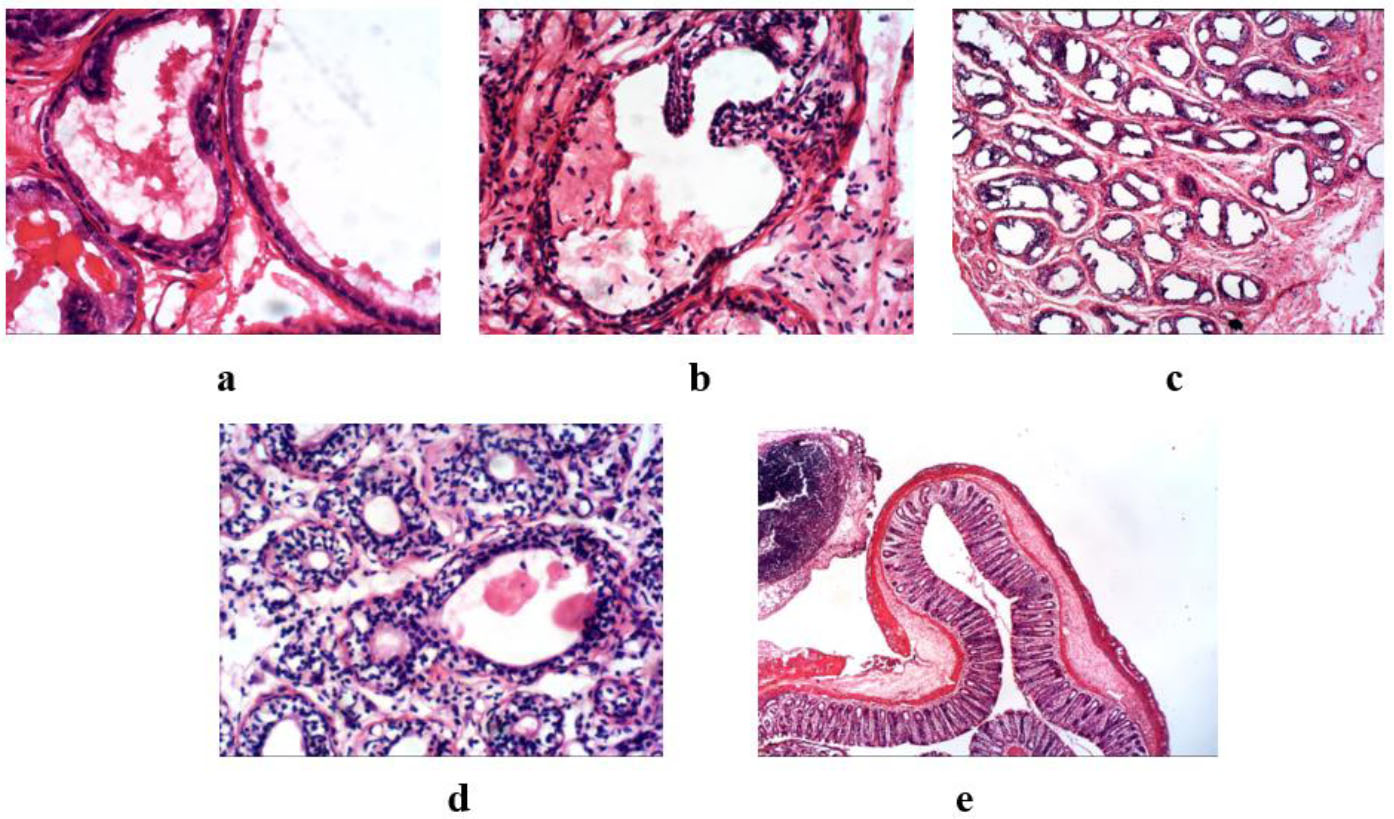
Photomicrograph of the histological evaluation of prostate tissue of MSG-induced prostate damage in rats.
The histopathology showed prostatic tissue composed of glands lined by low columnar epithelial cells with basal nuclei and supranuclear clear cytoplasm. (a) Control group: The prostate gland shows mild epithelial folds, contains secretion, and has no features of hyperplasia or atypia. (b) MSG-only: This group revealed prostatic tissue composed of glands lined by cuboidal epithelial cells and showed mild to moderate epithelial folds featuring glandular hyperplasia with some features of atypia. (c) ESEAM 50 mg/kg: Prostatic tissue is composed of glands lined by cuboidal epithelial cells with mild folds. Dense neutrophilic inflammatory infiltrates were present, but no hyperplasia or atypia were revealed. (d) ESEAM 100 mg/kg. Similar preservation of the glandular architecture with cuboidal epithelial lining and mild folds was seen. Cell infiltrations were noted, but no features of hyperplasia or atypia. (e) The finasteride group showed prostatic tissue composed of glands lined by cuboidal epithelial cells but no features of hyperplasia or atypia.
3.5. Molecular Docking Results
The molecular docking analysis of the ligands against the enzymes ALP, AST, ALT, and GGT, as shown in Table 6, reveals their potential inhibitory effects. The ligands with the highest binding affinity for all the enzymes are 552098, 5280450, 5364759, and 280342, particularly showing the highest binding to ALP (-157.696), AST (-140.326), ALT (-115.826), and GGT (-125.851), respectively. In contrast, the standard drug, 10970751, demonstrated the lowest binding affinities across all enzymes, particularly against ALP (-29.1069), suggesting a weaker inhibitory potential.
| Ligand CID | ALP | AST | ALT | GGT |
|---|---|---|---|---|
| 10944926 | -146.857 | -122.872 | -87.2935 | -108.168 |
| 5364759 | -154.658 | -137.115 | -115.826 | -115.826 |
| 5280450 | -155.779 | -140.326 | -106.307 | -106.307 |
| 586218 | -146.955 | -119.469 | -82.308 | -82.3081 |
| 552098 | -157.696 | -137.836 | -97.551 | -97.551 |
| 546976 | -137.837 | -122.497 | -85.9241 | -110.574 |
| 296573 | -135.499 | -146.052 | -95.601 | -129.073 |
| 280342 | -109.581 | -133.32 | -98.661 | -125.851 |
| 69340 | -140.73 | -125.463 | -69.340 | -117.761 |
| 13849 | -133.691 | -127.805 | -107.984 | -108.935 |
| 985 | -142.008 | -134.52 | -97.45 | -122.407 |
| 10970751 | -29.1069 | -93.9535 | -80.6544 | -80.6544 |
Ligands 13849 and 552098 exhibited strong binding to the ALP enzyme. Ligand 13849 hydrogen bonds with His 372, Glu 57, and Ser 56, suggesting their crucial role in enzyme binding, while 552098 interacts with Ser 56 via a hydrogen bond, strengthening its stability within the active site (Fig. 2). They also showed stearic interactions with some amino acid residues on both chains A and B, possibly contributing to their strong binding affinity and suggesting potential for effective inhibition of ALP activity, as shown in Table 7. The enzyme’s active site ligand stabilization may also be due to the interactions of Thr 413 and His 372 residues on both chains.
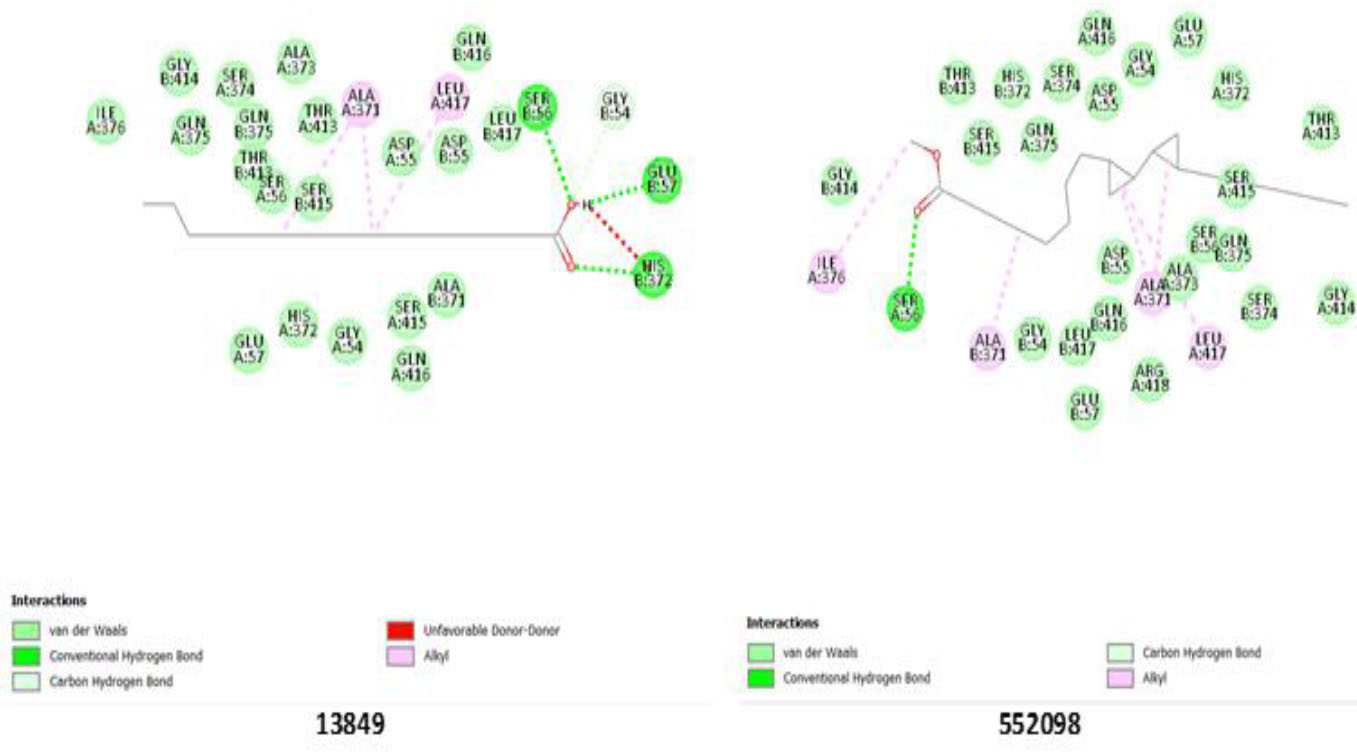
Binding pose showing interactions of ligands with amino acids in the active pocket of alkaline phosphatase enzyme.
| Ligands/Interactions | Hydrogen Bond with Amino Acid Residues | Other Interactions on both Chains A and B |
|---|---|---|
|
ALP 13849 552098 |
His 372(B); Glu 57(B); Ser 56(B) Ser 56(A) |
Glu 57(A); His 372 (A); Gly 54 (A and B); Ser 415(A and B); Ala 317(B); Gln 416(A and B); Ile 376(A); Gly 414 (B); Ser 374(A); Ala 373 (A); Gln 373 (A and B); Thr 413(A and B); Ala 371(A): Leu 417(A and B); Asp 55 (A and B); Ser 56 (A) Ile 376(A and B); Ala 371(B); Gly 54(A and B); Leu 417(A and B); Gln 416(A and B); Asp 55(A and B); Ala 373(A); Gln 375(A and B); Gly 414(A and B); Ser 374 (A and B); Arg 418(a); Glu 57(B); Thr 413(A and B); His 372(A and B); Ser 415(B) |
|
AST 10944926 5280450 |
His 181(A); Cys 183(A); Ala 184(A) His 181(A); Cys 183(A); Ala 216(A) |
Met 215(A); Tyr 217(A); Ala 182(A); Asp 214(A); Ala216(A); Gly 105(A); Trp 133(A); Tyr 255(A); Lys 250(A); Arg 378(A); Phe 352(A); Val 35(A); Leu 16(A); Asn 186(A); Ile 15(A); Ser 247(A); Arg 258(A); Ser 247(A); Ala 249(A); Thr 106(A); Leu 109(A); His 136(A) Thr 106(A); Arg 378(A); Lys 250(A); Tyr 255(A); Asn 186(A); Trp 133(A); Gly 105(A); Ala 216(A); Asp 214(A); Tyr 217(A); Met 215(A); Ala 182(A); Ala 184(A); His 185(A); His 136(A); Leu 109(A); Thr 106(A); Ala 249(A); Ser 247(A); Ile 15(A); Leu 16(A); Val 35(A); Gly 36(A) |
|
ALT 5364759 |
Lys 341(A) | Ser 350(A); Ser 338(a); Ala 187(A); Tyr 302(A); Val 301(A); Ile 191(A); Tyr 216(A); Ile 267(A); Asp 299(A): Glu 300(A); Gly 270(A); Asn 268(A); Pro 269(A); Asn 271(A); Tyr 219(A); Leu 218(A); Ser 188(A); Gly 186(A); Cys 347(A); Tyr 348(A); Gly 348(A) |
|
GGT 5280450 280342 |
Ser 452(B); Thr 381(B); Thr 399(B) Lys 562(B); Ser 82(A); Asn 401(B) |
Asn 401(B); Gln 507(B); Thr 475(B); Arg 107(B); Asn 431(B); Asp 422(B); Asp 423(B0; Arg 327(B); Asn 441(B); Phe 424(B); Ser 425(B); Leu 450(B); Gly 474(B); Ile 477(B); Met 453(B) Ala 382(B); Thr 381(B): Ala 472(B); Val 543(B); Tyr 404(B): Glu 420(B); Thr 399(B); Arg 107(A); Ser 451(B); Gly 473(B); His 81(A); Gly 474(B); Leu 402 (B); ile 477(B); Met 453(B); Ser 452(B); Asp 423(B) |
Ligands 10944926 and 5280450 showed significant binding to the AST enzyme. Both ligands hydrogen-bond to His 181(A), Cys 183(A), Ala 184(A), and Ala 216(A), indicating strong interactions within the catalytic pocket. Other steric interactions (Table 6) may contribute to enhancing ligand stabilization. The participation of residues Arg 378(A), Gly 105(A), and Trp 133(A) in the binding interaction suggests that these amino acids play a crucial role in ligand anchoring and enzyme inhibition. The potential inhibitory role of these ligands in AST function may be due to the consistency of hydrogen bonding and steric interactions highlighted in Table 6 and Fig. (3).
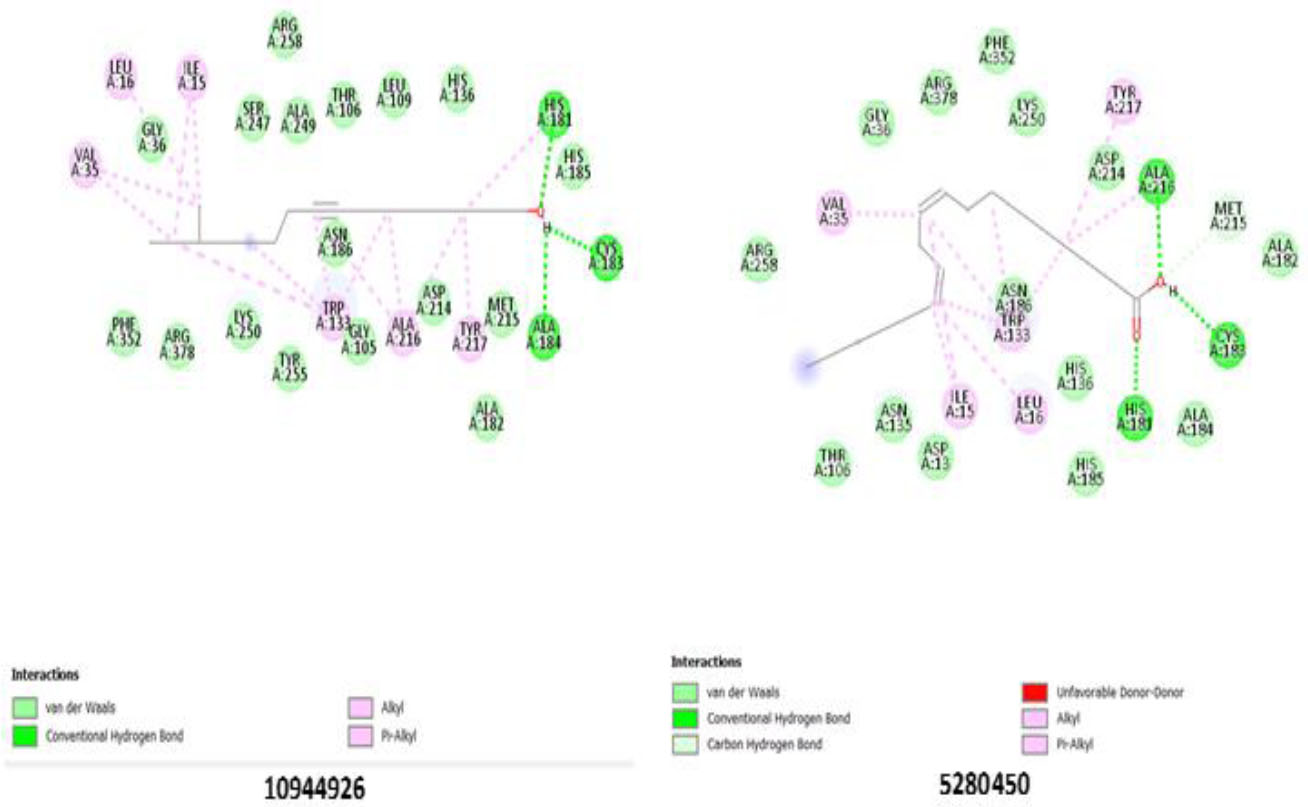
Binding pose showing interactions of ligands with amino acids in the active pocket of aspartate aminotransferase enzyme.
Confirming the strongest binding affinity for ALT enzyme, ligand 5364759 established a significant hydrogen bond with Lys 341(A). Moreover, multiple steric interactions with residues such as Ser 350(A), Ser 338(A), Ala 187(A), and Tyr 302(A) further supported its strong binding. The presence of interactions with Asn 268(A), Pro 269(A), and Asn 271(A) suggests that these residues play a role in stabilizing ligand binding. Ligand 5364759 may significantly inhibit ALT activity through interactions with catalytic residues, particularly Lys 341(A) (Fig. 4), making it a promising candidate for further evaluation.
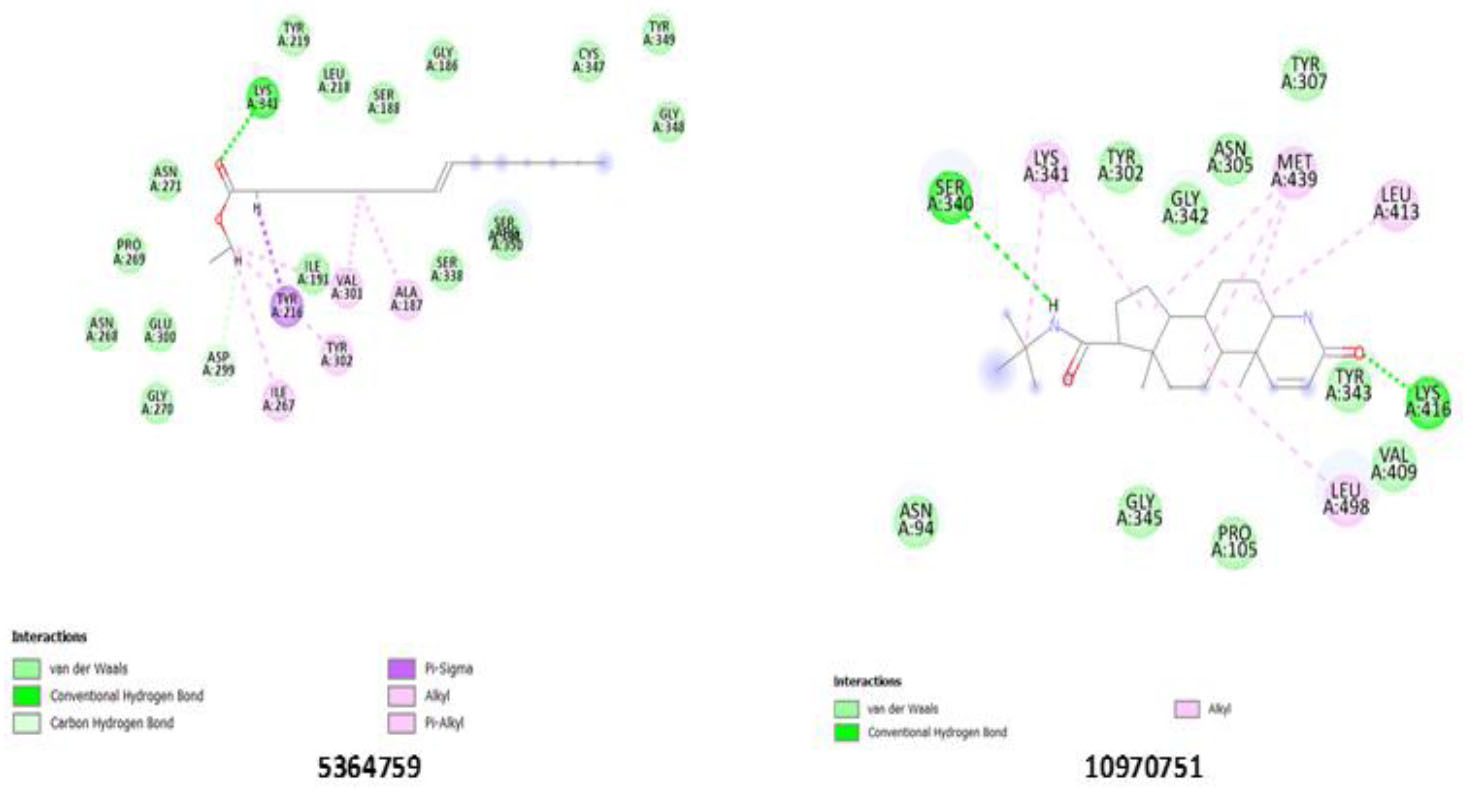
Binding pose showing interactions of ligands with amino acids in the active pocket of the Alanine aminotransferase enzyme.
Notable interactions with the GGT enzyme were displayed by ligands 5280450 and 280342. 5280450 hydrogen bonds with Ser 452(B), Thr 381(B), and Thr 399(B), strengthening its stability within the enzyme’s active site, while Lys 562(B), Ser 821(A), and Asn 401(B) strongly bond with 280342, emphasizing its potential inhibitory role (Fig. 5). The standard drug, 10970751, indicates a lower inhibitory potential compared to the ligands, as shown in Table 5, with the weakest binding affinity across all the enzymes. The tested ligands (13849, 552098, 10944926, 5280450, 5364759, and 280342) consistently outperformed ligand 10970751 in binding affinity, consistent with the 2D interactions of 10970751 with all the enzymes (Figs. S1-S3). Further validation may be needed to confirm their superior inhibitory properties and potential therapeutic applications via molecular dynamics simulation.
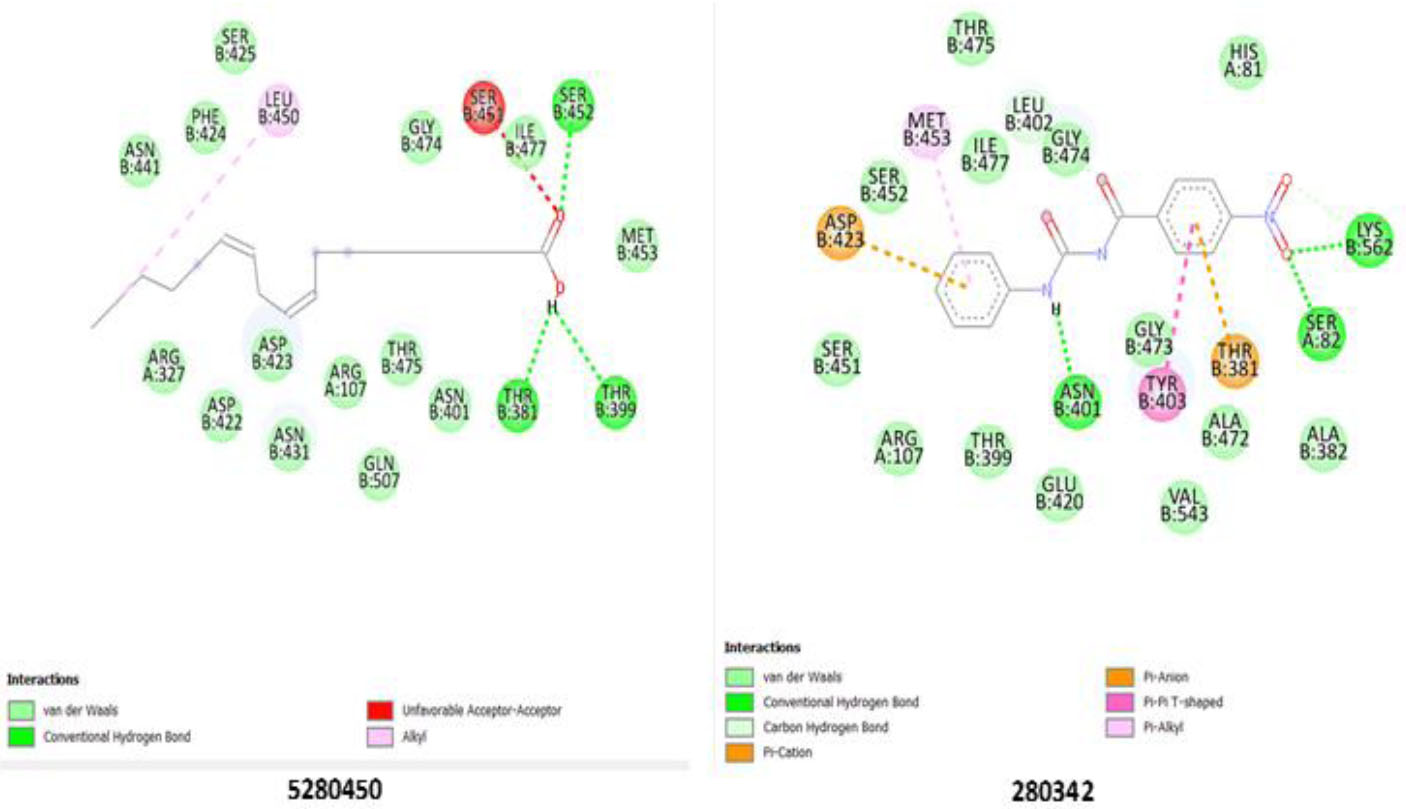
Binding pose showing interactions of ligands with amino acids in the active pocket of gamma-glutamyl transferase enzyme.
4. DISCUSSION
The results of this study demonstrate that intraperitoneal administration of ESEAM for 28 days suppressed the induction of MSG-induced prostate damage in Swiss Albino rats. Administration of ESEAM at doses of 50 and 100 mg/kg body weight conferred a significant increase in the hematological parameters of the MSG-treated groups as compared to the MSG-only group. This result agreed with the work of Eweka et al. [50], who reported that MSG adversely altered hematological indices, including lymphocyte and neutrophil counts, as well as other hematological parameters, with a corresponding increase in white blood cell counts, indicative of immune suppression and systemic toxicity. The reversal of this compromised immune status by ESEAM treatment is consistent with the study by Akpanabiatu et al. [51], who concluded that A. melegueta seed has the potential to ameliorate hematological indices in a rat model of benign prostatic hyperplasia (BPH). Also, MSG exposure led to a marked elevation of serum and hepatic marker enzymes (AST, ALT, ALP, and GGT), consistent with systemic inflammation and oxidative stress, both of which are recognized drivers of hepatocellular injury [52], consistent with previous reports of hepatocellular injury and prostate damage [44]. Oxidative stress, through ROS generation, can induce DNA damage and mutations, initiating malignant transformations [52]. Treatment with ESEAM significantly decreases the activity of these enzymes, indicating a hepatoprotective effect and mitigating ROS-associated damage, thereby interfering with pathways that predispose to malignancy. This finding is supported by the work of Kokou et al. [53], who reported that A. melegueta seed extract exerts hepatoprotective potential, likely mediated through antioxidant and anti-inflammatory mechanisms. Since ALP and GGT are clinically linked with prostate-related damage, the observed reduction suggests that ESEAM may confer protective benefits beyond hepatotoxicity, potentially modulating pathways relevant to prostate pathology.
The study also revealed that kidney indices (urea, uric acid, and creatinine) increased significantly in the MSG-treated groups compared with the control group. Although ESEAM and finasteride did not completely normalize these parameters, both treatments attenuated the MSG-induced elevations, suggesting a nephroprotective effect. In addition, MSG exposure significantly elevated serum and prostate markers (PSA, LH, TACP, and PACP) when compared with the control group. These elevated levels are indicative of androgen dysregulation, a hallmark of prostate disease. PSA is transcriptionally regulated by the androgen receptor (AR), which plays a central role in tumor initiation and progression [54]. Increased levels of these markers are consistent with the development of prostate damage and agree with the work of Ito et al [55] who reported that serum and marker concentrations of 4.0 ŋg/mL and above are indicative of prostate malignancy or damage. ESEAM treatment ameliorated these changes, thereby supporting its protective role against MSG-induced prostate alterations. This is consistent with previous findings that plant phytochemicals from Aframomum melegueta may exert AR-modulatory or inhibitory effects [56]. However, while elevated prostate markers and histopathology changes suggest prostate damage in this model, further studies incorporating definitive cancer biomarkers are necessary to exclude benign conditions.
Molecular docking analysis further revealed the inhibitory potential of selected ligands from ESEAM (552098, 5280450, 5364759, and 280342) against key enzymes, specifically ALP, AST, ALT, and GGT. While AST and ALT are the major circulating enzymes in the serum and are traditionally used as indicators of hepatocellular damage, emerging evidence suggests that their dysregulation may also accompany malignant progression in several cancers, including pancreatic and breast cancer [57-59]. γ-Glutamyltransferase (GGT), an oxidative stress biomarker, has been linked to poor survival in various malignancies in elevated conditions [60], whereas ALP is frequently elevated in prostate pathologies and bone metastasis [61]. The interactions observed in the docking results, including strong binding affinities, consistent hydrogen bonding, and steric interactions, suggest that these phytochemicals may contribute to therapeutic effects by modulating enzymatic activity associated with tissue damage in prostate disease, even though docking studies provide only predictive insights.
CONCLUSION
Ethanol seed extract of Aframomum melegueta (ESEAM) intraperitoneal administration has been revealed in this study to significantly alleviate the adverse effects of MSG-induced prostate damage in rats. In addition to improving hematological parameters, administration of ESEAM (at doses of 50 and 100 mg/kg body weight) effectively reversed MSG-induced prostate damage, enhanced serum and liver marker profiles, and suggested its potential as a protective agent against MSG-induced toxicity. Also, ESEAM administration positively influenced kidney indices, reflecting its potential renoprotective effects. Molecular docking studies revealed that the tested ligands (13849, 552098, 10944926, 5280450, 5364759, and 280342) exhibited strong binding interactions with the active sites of the enzymes ALP, AST, ALT, and GGT, suggesting their potential to inhibit enzyme activity. The highest binding affinities to ALP and ALT were observed with ligands 552098 and 5364759, compared with the standard drug, indicating their promising potential for enzyme inhibition. This shows the potential of ESEAM as a therapeutic agent for the management of prostate damage, with promising molecular interactions and its efficacy demonstrated through hematological, hepatic, and renal improvements. Further studies, including molecular dynamics simulations and network pharmacology, are warranted to validate these findings and explore the clinical applications of ESEAM and its active ligands.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contribution to the paper as follows: S.A., N.K.: Study conception and design; Q.H.: Methodology; F.A.: Investigation; A.N., U.G.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| ALT | = Alanine Aminotransferase |
| ALP | = Alkaline Phosphatase |
| AST | = Aspartate Aminotransferase |
| AR | = Androgen Receptor |
| BPH | = Benign Prostatic Hyperplasia |
| DHT | = Dihydrotestosterone |
| EDTA | = Ethylene Diamine Tetraacetic Acid |
| ESEAM | = Ethanol Seed Extract of Aframomum Melegueta |
| GC-MS | = Gas Chromatography-Mass Spectrometry |
| GGT | = Gamma-Glutamyltransferase |
| IL-6 | = Interleukin-6 |
| LH | = Luteinizing Hormone |
| MSG | = Monosodium Glutamate |
| MVD | = Molegro Virtual Docker |
| MW | = Molecular Weight |
| PACP | = Prostatic Acid Phosphatase |
| PSA | = Prostate-Specific Antigen |
| ROS | = Reactive Oxygen Species |
| RT | = Retention Time |
| TACP | = Total Acid Phosphatase |
| TNF-α | = Tumor Necrosis Factor-α |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved and conducted in accordance with the ethical procedures and policies of the Animal Care and Use Committee of Kwara State University, Malete, Nigeria, and an approval number (KWASU/CR&D/REA/2024/050) was assigned.
HUMAN AND ANIMAL RIGHTS
All animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals.
This study adheres to internationally accepted standards for animal research and follows the 3Rs principle. The ARRIVE guidelines were employed for reporting experiments involving live animals, promoting ethical research practices.
AVAILABILITY OF DATA AND MATERIALS
All the data and supporting material is available within the article.
ACKNOWLEDGEMENTS
Declared none.


